BioNTech CEO: Third dose of coronavirus vaccine necessary after a year, followed by annual booster shots
05/11/2021 / By Arsenio Toledo
The chief executive officer of German pharmaceutical company BioNTech believes people need to take a third dose of the Wuhan coronavirus (COVID-19) vaccine his company created with Pfizer after a year, and annual booster shots every 12 to 18 months after that.
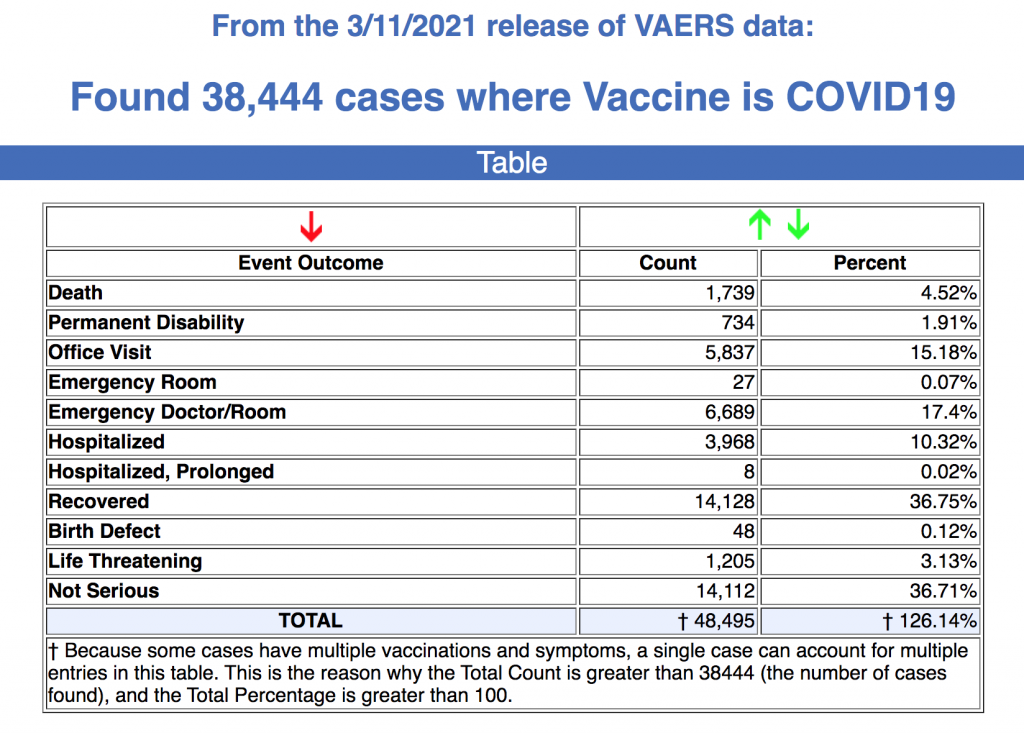
BioNTech CEO Dr. Ugur Sahin said, according to studies, the efficacy of the BioNTech-Pfizer coronavirus vaccine drops from 95 percent to 91 percent six months after the second dose. This is why his company believes people should be given a third dose nine to 12 months after the first vaccine dose.
“Accordingly, we need a third shot to get the vaccine protection back up to almost 100 percent again,” said Sahin. “And then I expect it will probably be necessary to get another booster every year or perhaps every 18 months.” (Related: PLAGUE PROFITS: Pfizer executive admits the company wants to profit off vaccines and will soon raise prices.)
BioNTech co-founder and Chief Medical officer Dr. Ozlem Tureci agreed with Sahin’s assessment. She told CNBC on an April 21 interview that as immunity against the coronavirus decreases over time, a third vaccine dose is necessary to maintain the vaccine’s effectiveness.
She compared the need to take the coronavirus vaccine annually to the flu vaccine.
“It is the nature of immune responses that after they have been induced they spike and stay for a time,” said Tureci during the interview. “But with time immune responses wane and we see indications for this in the induced, but also in the natural response against SARS-CoV-2.”
Sahin and Tureci’s comments regarding a third dose of the coronavirus vaccine come after Pfizer CEO Albert Bourla said a booster dose will likely be necessary for people within 12 months after becoming fully vaccinated.
BioNTech claims vaccine effective against all existing coronavirus strains
BioNTech is confident that its coronavirus vaccine will be able to fight off the many variants of COVID-19.
Sahin said his company has tested its vaccine against more than 30 coronavirus variants. He claimed the vaccine showed a “promising response” against most of the variants, while the vaccine “remained sufficient” against the other variants where the response was slightly diminished.
Furthermore, Sahin believes the BioNTech-Pfizer coronavirus vaccine can protect people against the new variant first detected in India, but that the company was still conducting its investigation.
“But the Indian variant has mutations that we have previously investigated and against which our vaccine also works, so I am confident there, too,” he claimed.
Sahin explained that, the way the BioNTech vaccine works gives it “two points of attack” against the virus. It supposedly stimulates the production of antibodies while at the same time prompts the body’s T-cells to attack the virus.
“The vaccine is quite cleverly constructed, and the bulwark will hold,” said the CEO. “I’m convinced of that. If the bulwark needs to be strengthened again, we’ll do so. I’m not worried.”
BioNTech and Pfizer are working with their manufacturers to ramp up production of the coronavirus vaccine as worldwide demand for it still far outstrips their supplies.
“When we started 2021, our goal was to produce 1.3 billion doses,” said Sahin. “And now, we’ve increased that to three billion doses.”
The company is in talks to open production facilities all over South America, Asia and Africa. It is also considering issuing special licenses to certain manufacturers to boost the production of the vaccine.
Sahin and Tureci, who are married, founded BioNTech in 2008. They used to focus most of their efforts on cancer research before deciding to pivot to creating a coronavirus vaccine in early 2020. It reached out to Pfizer, which had the resources and the expertise to conduct the large-scale clinical trials necessary to validate a vaccine’s supposed effectiveness.
Learn more about coronavirus vaccines by reading the latest articles at Vaccines.news.
Sources include:
Tagged Under: Big Pharma, BioNTech, booster shot, coronavirus, coronavirus vaccine, coronavirus variants, covid-19, immunization, mutation, pandemic, Pfizer, third dose, vaccines
RECENT NEWS & ARTICLES
Vaccines.News is a fact-based public education website published by Vaccines News Features, LLC.
All content copyright © 2018 by Vaccines News Features, LLC.
Contact Us with Tips or Corrections
All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.