Surgical tools from Johnson & Johnson RECALLED due to increased risk of injury and death
07/18/2023 / By Zoey Sky
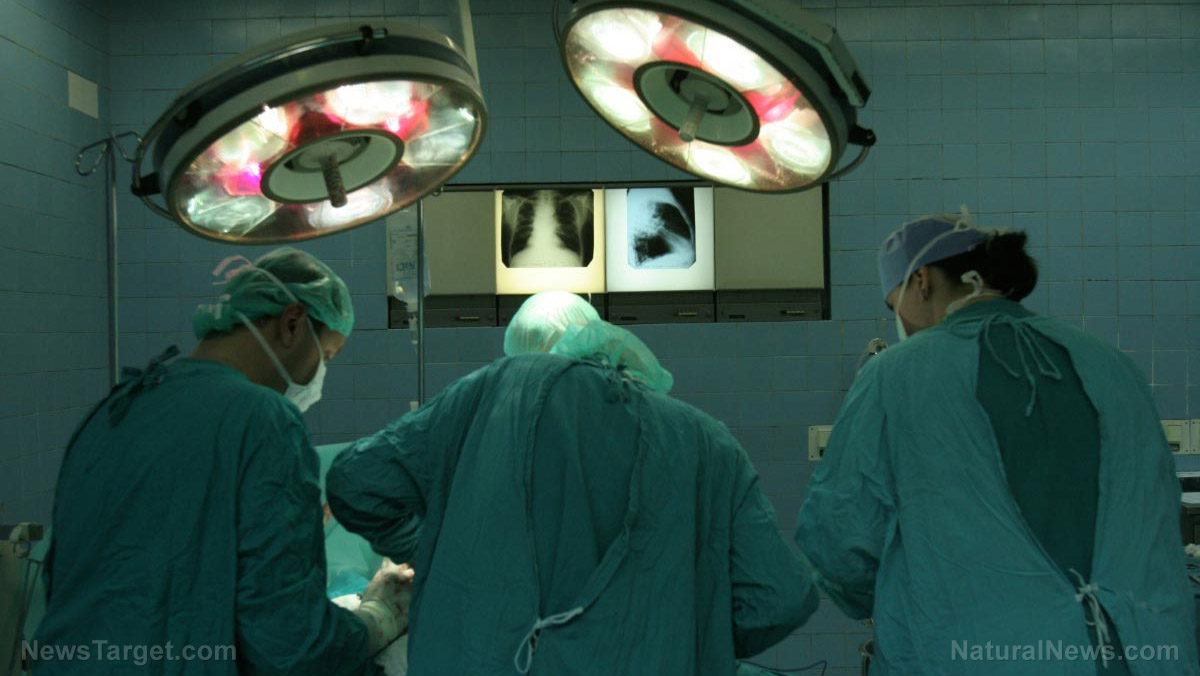
Surgical tools from Johnson & Johnson used to make incisions and stop bleeding during surgery have been recalled because of the increased risk of injury and death, according to reports.
The Food and Drug Administration (FDA) announced the recall in a statement, adding that it received reports of young and older patients suffering burn injuries when the J&J surgical tools were used on them. The regulator classified the recall under the “most serious” category.”
The tools, which were sold under the brand names MEGA 2000 Reusable Patient Electrodes and MEGA SOFT Reusable Patient Electrodes, were recalled by Megadyne Medical Products, a Utah-based company. Megadyne was acquired in 2017 by Ethicon Endo-Surgery, a unit of J&J MedTech.
The company received reports of at least 63 injuries among patients but no deaths related to the surgical tools. Megadyne reported that it will continue to reassess the root cause for patient burns.
The recalled surgical tools are soft pads that conduct an electric current and heat the patient’s skin. The pads are used to make incisions and stop bleeding during surgery.
Aside from J&J, other companies manufacture these tools, which are commonly used in different types of surgeries.
An investigation revealed that in some cases, the tools were used incorrectly, claimed J&J MedTech. (Related: FDA warns against using COVID-19 at-home test due to bacterial contamination.)
In June, the manufacturer recalled 21,200 units of the MEGA 2000 Reusable Patient Electrodes and MEGA SOFT Reusable Patient Electrodes, which were distributed between March 11, 2021, and May 9, 2023.
J&J MedTech also reported that the products are still available. It issued notices in June to healthcare professionals to follow proper instructions for the use of the pads.
It is still unknown how severe the burn injuries were and how exactly the burns occurred during surgery.
J&J vaccine also recalled due to blood clot concerns
The surgical tools aren’t the first products recalled by the FDA over serious concerns. Back in June, it also revoked authorization of J&J’s Wuhan coronavirus (COVID-19) vaccine. The decision followed a pause in the shot’s use over blood clot concerns, which saw a significant decline in the demand for it.
The move was not unexpected because Janssen, J&J’s parent company, requested that federal regulators at the FDA withdraw authorization for its vaccine after it was revealed that the last tranche of doses, totaling at least 12.5 million doses, had already expired.
By the third year of the COVID-19 pandemic, an estimated 231 million Americans have received either one dose of the J&J vaccine or two doses of an mRNA vaccine from either Pfizer or Moderna.
Less than a month after the J&J vaccine was granted emergency use authorization from the FDA, it was embroiled in controversy over a small but growing number of severe blood clotting disorders called thrombosis with thrombocytopenia syndrome (TTS) in people who received the vaccine.
TTS is a severe illness characterized by thrombosis formation, or blood clots, combined with thrombocytopenia, a severely low platelet count that hinders the body’s ability to stop a wound from bleeding.
Symptoms of TTS, also known as vaccine-induced prothrombotic immune thrombocytopenia (VIPIT) or vaccine-induced immune thrombotic thrombocytopenia (VITT), typically occur between four and 42 days after vaccination.
Visit DangerousMedicine.com for more stories like this.
Watch the video below to know more about the pause on the use of Johnson and Johnson COVID-19 vaccines.
This video is from The Prisoner channel on Brighteon.com.
More related stories:
Lawsuit reveals toxic oil used in IVF destroyed California couple’s embryos.
FDA probing cases of FOOD POISONING from Lucky Charms breakfast cereal.
FDA announces voluntary recall of contaminated eye drops that could blind people.
J&J offers almost $9B to settle lawsuits alleging its talc-based baby powder caused cancer.
Sources include:
Submit a correction >>
Tagged Under:
Big Pharma, J&J MedTech, medical violence, MEGA 2000 Reusable Patient Electrodes, MEGA SOFT Reusable Patient Electrodes, Megadyne Medical Products, pharmaceutical fraud, Product recall, products, vaccine damage, vaccine injury, vaccines
This article may contain statements that reflect the opinion of the author
RECENT NEWS & ARTICLES
Vaccines.News is a fact-based public education website published by Vaccines News Features, LLC.
All content copyright © 2018 by Vaccines News Features, LLC.
Contact Us with Tips or Corrections
All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.